(Eagan, MN) — April 27, 2022 — Sonex Health today announced the full U.S. commercial launch of its UltraGuideTFR device, which is used in conjunction with real-time ultrasound guidance to perform trigger finger release (TFR) procedures. This follows Sonex Health’s successful limited-market release earlier this year in which more than 100 patients were treated using the device. Real-time ultrasound guidance enables physicians to use a minimally invasive technique while performing TFR through a small incision, giving most patients the ability to resume activities within days versus weeks with traditional surgeries.
“The full launch of UltraGuideTFR enables us to offer more clinicians safe and effective ways to treat common painful upper extremity conditions using minimally invasive techniques,” said Sonex Health CEO Bob Paulson. “Our devices, UltraGuideTFR and UltraGuideCTR to treat carpal tunnel syndrome, both harness the power of ultrasound, and were designed to deliver patient and clinician satisfaction by reducing invasiveness, cost, and recovery time.”
“Trigger finger” syndrome is the common name for stenosing tenosynovitis, a painful condition caused by a progressive restriction of normal tendon motion by the A1 pulley, interfering with the ability to bend and straighten the fingers.
UltraGuideTFR is a single-use, hand-held device developed by the physician co-founders of Sonex Health – Dr. Darryl Barnes and Dr. Jay Smith – both of whom previously practiced at Mayo Clinic and founded the Institute of Advanced Ultrasound Guided Procedures, which focuses on product innovation, clinical research, and educating physicians on how to hone their musculoskeletal ultrasound skills. UltraGuideTFR is indicated for the treatment of trigger finger and is designed to incise the tendon sheath and A1 pulley through a single incision.
Real-time ultrasound guidance allows treating physicians to visualize the critical anatomy prior to and during the tendon sheath incision, as well as probe the tendon sheath and A1 pulley to confirm an adequate release. Previous studies of TFR procedures using ultrasound have shown they can be performed in procedure rooms or office settings utilizing local anesthesia and have been associated with reduced pain and rapid recovery.
“Real-time ultrasound guidance allows me to see all critical anatomy prior to making an incision, which enables me to guide the device while performing the procedure through a much smaller incision,” said Dr. Richard Schaefer of SSM Health in Fond du Lac, Wis., who was part of the limited market release. “Because this technique is less invasive, patients recover sooner and return to their normal activities and jobs much faster than they would with traditional TFR surgery.”
During the limited market release, physicians provided user experience feedback on 100 TFR procedures using UltraGuideTFR.
Results included:
- Ninety-seven (97) percent of the procedures were performed in the office, procedure room or ambulatory surgery center under local anesthesia.
- There were no complications, and a complete release of the tendon sheath and A1 pulley was confirmed for all procedures.
- Physicians rated the performance of the device and procedure on a scale of 1-5 (1 = strongly disagree and 5 = strongly agree) with the following results:
- Easy to use = 4.98 (n=100)
- Easy to perform the procedure = 4.98 (n=100)
- Will use with future TFR patients = 4.98 (n=100).
“Trigger finger can interfere with daily life, especially for those who rely on using their hands for their livelihoods. Using ultrasound allows me to perform TFR procedures in my office under local anesthesia,” said Dr. Douglas Hoffman of Essentia Health SMMC in Duluth, Minn., who also participated in the TFR limited market release. “Patients really appreciate the convenience of avoiding the operating room for this procedure.”
Trigger finger syndrome affects almost nine million Americans and results in 350,000 surgeries every year. While traditional surgery to treat TFR can remedy the condition, the surgery may result in large, and sometimes painful scars and a long recovery. TFR procedures using the UltraGuideTFR can be performed through a small incision, which can typically be closed with an adhesive strip or bandage instead of sutures.
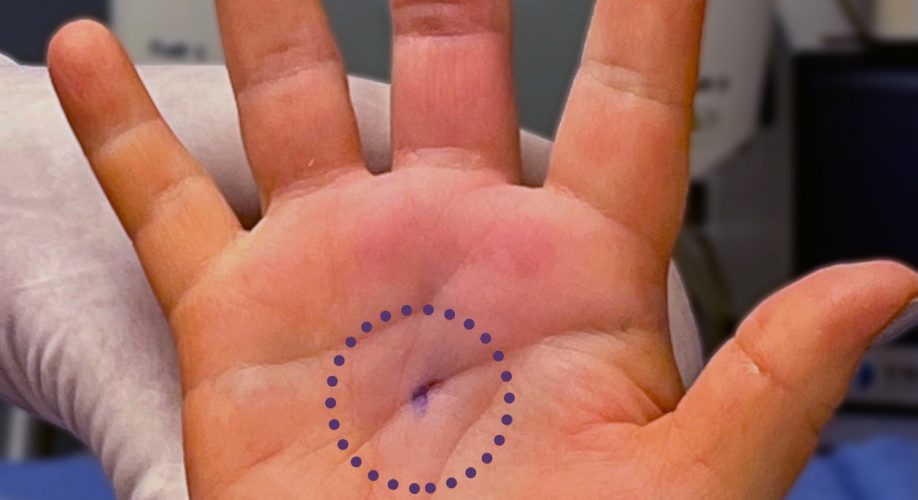
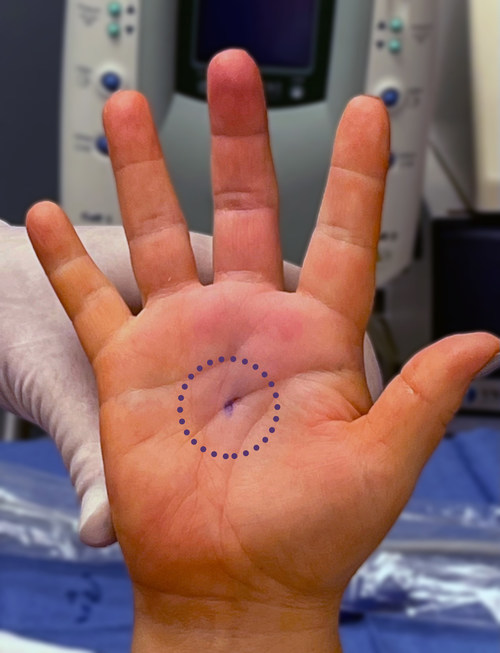
Dr. Richard Schaefer of SSM Health in Fond du Lac, Wis., shows the small incision of his patient’s hand immediately after a trigger finger release with Sonex Health’s UltraGuideTFR and real-time ultrasound guidance.
ABOUT SONEX HEALTH
Founded in 2014, Sonex Health’s mission is to be the world leader in ultrasound guided surgery by delivering physicians innovative therapies that reduce invasiveness, improve safety, and reduce the cost of care. With a strong focus on entrapment neuropathy, Sonex Health’s first proprietary technology — developed by Dr. Darryl E. Barnes and Dr. Jay Smith at the world-renowned Mayo Clinic — is UltraGuideCTR (formerly referred to as SX-One Micro-Knife), which may be utilized with or without ultrasound guidance to perform carpal tunnel release. Sonex Health’s second proprietary technology is UltraGuideTFR, for the treatment of trigger finger, also known as stenosis tenosynovitis.
For information about Sonex Health, UltraGuideCTR, and UltraGuideTFR visit www.sonexhealth.com.
ABOUT THE INSTITUTE OF ADVANCED ULTRASOUND GUIDED PROCEDURES
Founded in 2018 to support the Sonex Health mission and clinical excellence, the Institute of Advanced Ultrasound Guided Procedures is focused on innovation supported by robust clinical research and world-class professional education and training that transforms the treatment experience for patients, providers and payers. For more information about the Institute visit www.sonexhealth.com/educational-institute.